
Caplin Point Laboratories Ltd – The Art of Uncompromising
Established in 1990 to manufacture a range of ointments, creams and other external applications, Caplin Point Laboratories Ltd. is now a fast growing fully integrated global pharmaceutical company with a dominant presence in Latin America and Francophone Africa. The company has established it’s position as one of the leading suppliers of pharmaceuticals in these regions, with over 4000 product licenses across the globe. It has a growing presence in the regulated markets such as USA and EU as well. The company is present across all critical and value driven spots on the pharmaceutical value chain right from APIs to Finished Formulations; Research & Development; Clinical Research; Contract Research Organization (CRO); Marketing & Distribution; Online Platform for Pharmacy Automation; and the recently added Key Starting Material (KSM) manufacturing capabilities. Caplin has presence across 36 therapeutic areas and 650+ formulations products as of 31 March 2023.
Products and Services
Caplin’s product list comprises of tablets, capsules, injections (liquid & lyophilized, pre-filled syringes), ophthalmic, softgel capsules, liquid orals, ointments, cream and gels, powder for injections, suppositories and ovules, pre-mix bags and inhalers and sprays. The company has different categories of products based on geographical market segments – regulated and emerging markets.
Subsidiaries: As of FY23, the company had 11 subsidiaries (including step down subsidiaries) and 1 associate company.
Key Rationale
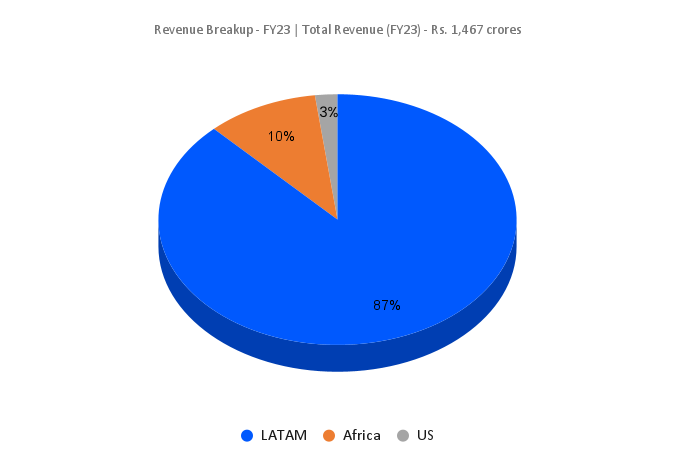
- Consistently growing “Emerging Markets” Business – The company is following a differentiated approach by focusing majorly on the semi regulated markets of LatAm and Africa, which contributes the major portion of overall sales. Over the year’s Caplin has established its presence in these areas with its distribution networks. It aims to expand its presence in the bigger markets of Mexico, Brazil, Argentina, Colombia etc. The focus is also on expanding the business in US markets through injectables which offers immense growth opportunities.
- Expansion plans – The company is establishing API facility in Vizag, that is largely a backward integration move aiming to establishing its position in generic injectable/ophthalmic products in the Regulated space. Additionally, oncology API site construction is under progress at Chennai. In terms of approvals there are 13 approvals under review with FDA. This comprises of ophthalmic products, four premixed bags, which is a niche area without much competition and injectable products of which one is a complex emulsion. Oncology products (tablet, capsule & injectable) and softgel are also two niche segments that company is focusing on. During FY23, the company developed pipeline of a new line of products – Oncology. Phase 1 of the oncology facility will have oral solid dosages and Phase 2 would be manufacturing injectables.
- Q2FY24 – During the quarter, revenue increased by 14% to Rs.410 crores compared to the Rs.359 crores of Q2FY23. On account of outsourced oncology sales, increase in soft gels sales and improved revenue from Caplin Steriles Ltd. (a material subsidiary), EBITDA and PAT have improved. EBITDA increased by 29% from Rs.106 crores of Q2FY23 to Rs.137 crores of Q2FY24. The company reported a net profit of Rs.116 crores during the quarter, an growth of 26% compared to the same period previous year. The EBITDA and net profit margin are 33% and 28% respectively.
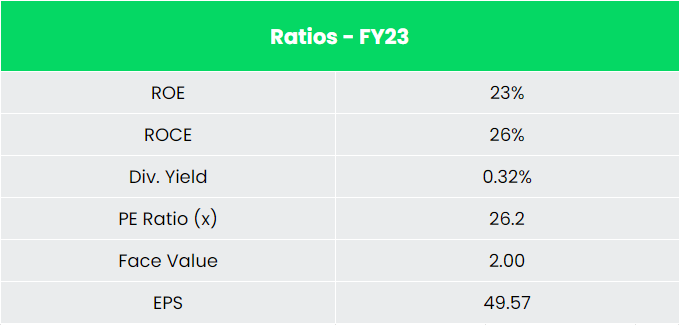
- Financial performance – The company has generated revenue and PAT CAGR of 22% and 21% over the period of 5 years (FY18-23). Average 5-year ROE & ROCE is around 28% and 32% for FY18-23 period. The company has strong balance sheet with zero debt in its balance sheet. Reducing cost by an asset light business model is a strategy the company is implementing by outsourcing from India and China for products that are pure vanilla generics where the overhead is very low.

Industry
Serving across 200+ countries, the Indian Pharmaceuticals industry plays a prominent role in the global pharmaceuticals requirements. India is one of the biggest suppliers of low-cost vaccines in the world and is the largest provider of generic medicines globally, occupying a 20% share in global supply by volume. India also has the highest number of US-FDA compliant pharma plants outside of USA and is home to more than 3,000 pharma companies with a strong network of over 10,500 manufacturing facilities as well as a highly skilled resource pool. There are 500 API manufacturers contributing about 8% in the global API Industry. India is the largest supplier of generic medicines manufacturing about 60,000 different generic brands across 60 therapeutic categories and accounts for 20% of the global supply of generics. The pharmaceutical industry in India is expected to reach $65 Bn by 2024 and to $130 Bn by 2030. India is rightfully known as the “pharmacy of the world” due to the low cost and high quality of its medicines.
Growth Drivers
100% Foreign Direct Investment (FDI) in the pharmaceutical sector is allowed under the automatic route for greenfield pharmaceuticals. 100% FDI in the pharmaceutical sector is allowed in brownfield pharmaceuticals; wherein 74% is allowed under the automatic route and thereafter through the government approval route. The Ministry’s scheme “Strengthening of Pharmaceutical Industry (SPI)” with a total financial outlay of US$ 60.9 million (Rs. 500 crore) extends support required to existing pharma clusters and MSMEs across the country to improve their productivity, quality and sustainability. As per Union Budget 2022-23 Rs. 3,201 crore (US$ 419.2 million) has been set aside for research and Rs. 83,000 crore (US$ 10.86 billion) has been allocated for the Ministry of Health and Family Welfare.
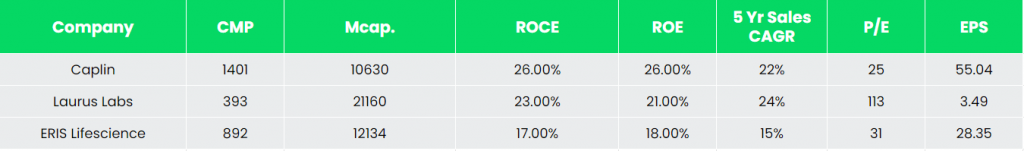
Competitors: Laurus Labs, ERIS Lifescience
Peer Analysis
Among the above competitors, with a reasonably steady revenue growth, Caplin has better return ratios and robust earnings potential, indicating the company’s financial stability and its efficiency to generate income and returns from the invested capital.
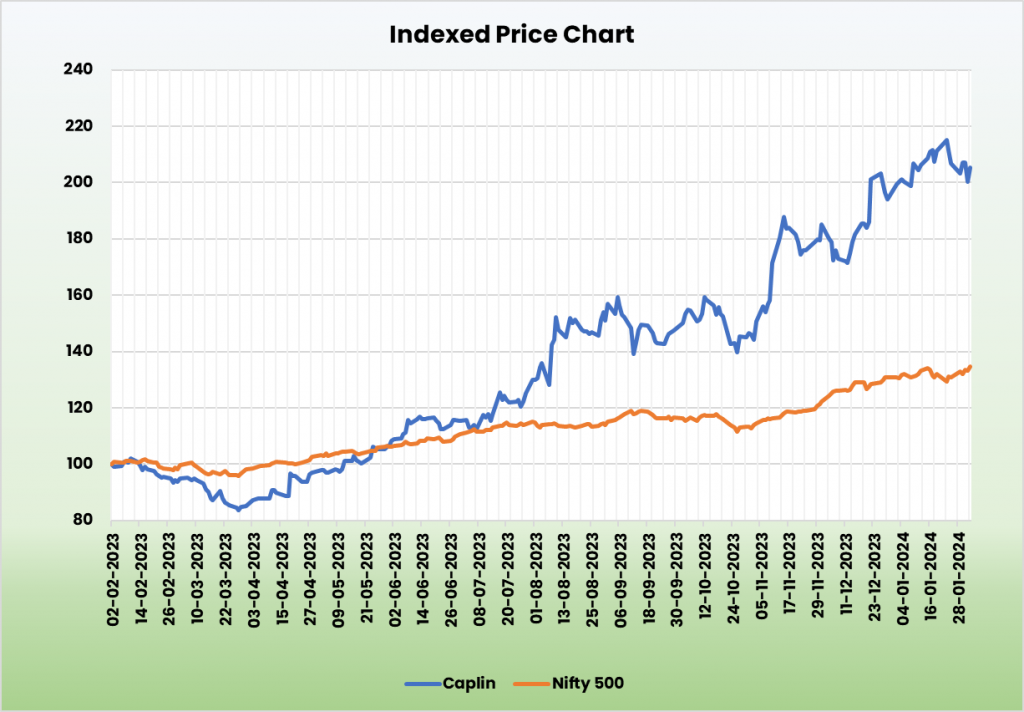
Outlook
The choice of growth paths of exports to lesser penetrated and relatively unregulated markets has proved to be a path of success for the Caplin. There are plans afoot to enter Canada, Australia, China and Brazil shortly and we expect the company to gain momentum in these markets in the long term. It is continuously working on R&D and adding more segments to the product portfolio. Initiatives of forward and backward integration such as outsourcing, having own distribution channels, following an asset light model, warehouses near customers and new API plants reduces supply chain challenges, cash flow issues and improves margins. Expansion plans are funded from internally generated cash with a commitment to maintain a debt-free capital structure.
Valuation
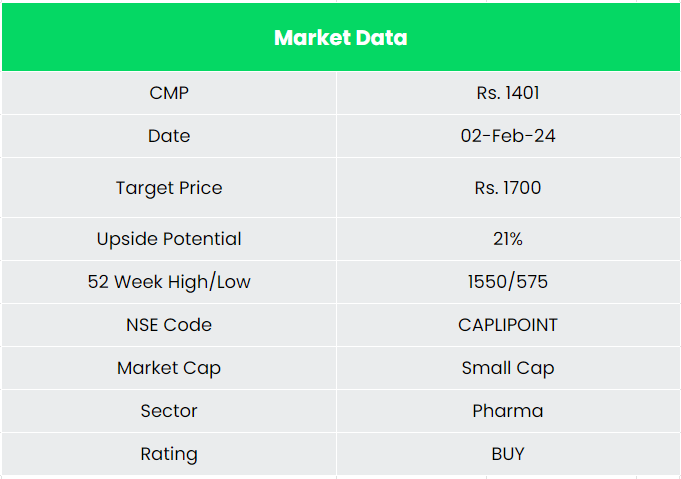
We believe Caplin Point Laboratories Ltd is in a position for robust growth in the coming years. It’s growing market share in the existing business and upcoming projects the company has in pipeline places it in a position for a strong growth potential. We recommend a BUY rating in the stock with the target price (TP) of Rs. 1700, 21x FY25E EPS.
Risks
- Forex Risk – The company has significant operations in foreign markets and hence is exposed to forex risk. Any unforeseen movement in the forex market can adversely affect the company.
- Regulatory risk – The industry is highly susceptible to regulatory changes, and this might result in limitation/ban of certain products, affecting revenue. The operations are exposed to regulatory risk, including scrutiny by regulatory agencies like the USFDA which might lead to restrictions/ban in products, affecting company operations.
- Geopolitical crisis – The company has operations in certain geographical segments which are sensitive to geopolitical outbreaks. Any unprecedented movements in these areas might impact turnover.
Other articles you may like
Post Views:
212


